Deterministic modeling and stochastic simulation of poly-alkoxylation reactions
Riccardo Tesser, Elio Santacesaria
Journal of the Indian Chemical Society Volume 98, Issue 4, April 2021, 100054 https://doi.org/10.1016/j.jics.2021.100054
Abstract
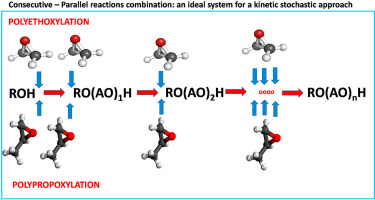
The main scope of this work is to show the feasibility and the advantage of using a stochastic approach to describe the poly-alkoxylation kinetics of different substrates. For this purpose, the reactions of ethylene and propylene oxides with respectively ethylene glycol, 1-octanol, and 2-octanol were considered. Two kinetic models were used for interpreting all the kinetic runs available in the literature, one deterministic and another one stochastic, for a useful comparison between the two different approaches. As the adopted reaction mechanism, rate laws, and related kinetic parameters were the same for both the kinetic models, the obtained results for what concerns the substrate consumption, and the oligomers distribution profiles were the same in both cases. In the case of the stochastic kinetic approach, the calculations must be made on a small volume of the reacting mixture containing a sufficiently high number of molecules that is suitable for the statistical analysis but as small as possible for reducing the calculation time. The calculations made have allowed individuating this optimal volume. This study is propaedeutic to the application of a stochastic kinetic approach to the study of ethylene-propylene oxides copolymerization that cannot be faced with a deterministic model for the extremely long or impracticable calculation time due to the great number of material balance differential equations that must be integrated.