Maryam Hmoudah, Amjad El-Qanni, Saqr Abuhatab, Nedal N. Marei, Amer El-Hamouz, Belal J. Abu Tarboush, Ihab H. Alsurakji, Hanaa M. Baniowda, Vincenzo Russo & Martino Di Serio
Environmental Science and Pollution Research volume 29, pages77992–78008 (2022)
https://doi.org/10.1007/s11356-022-21368-7
Abstract
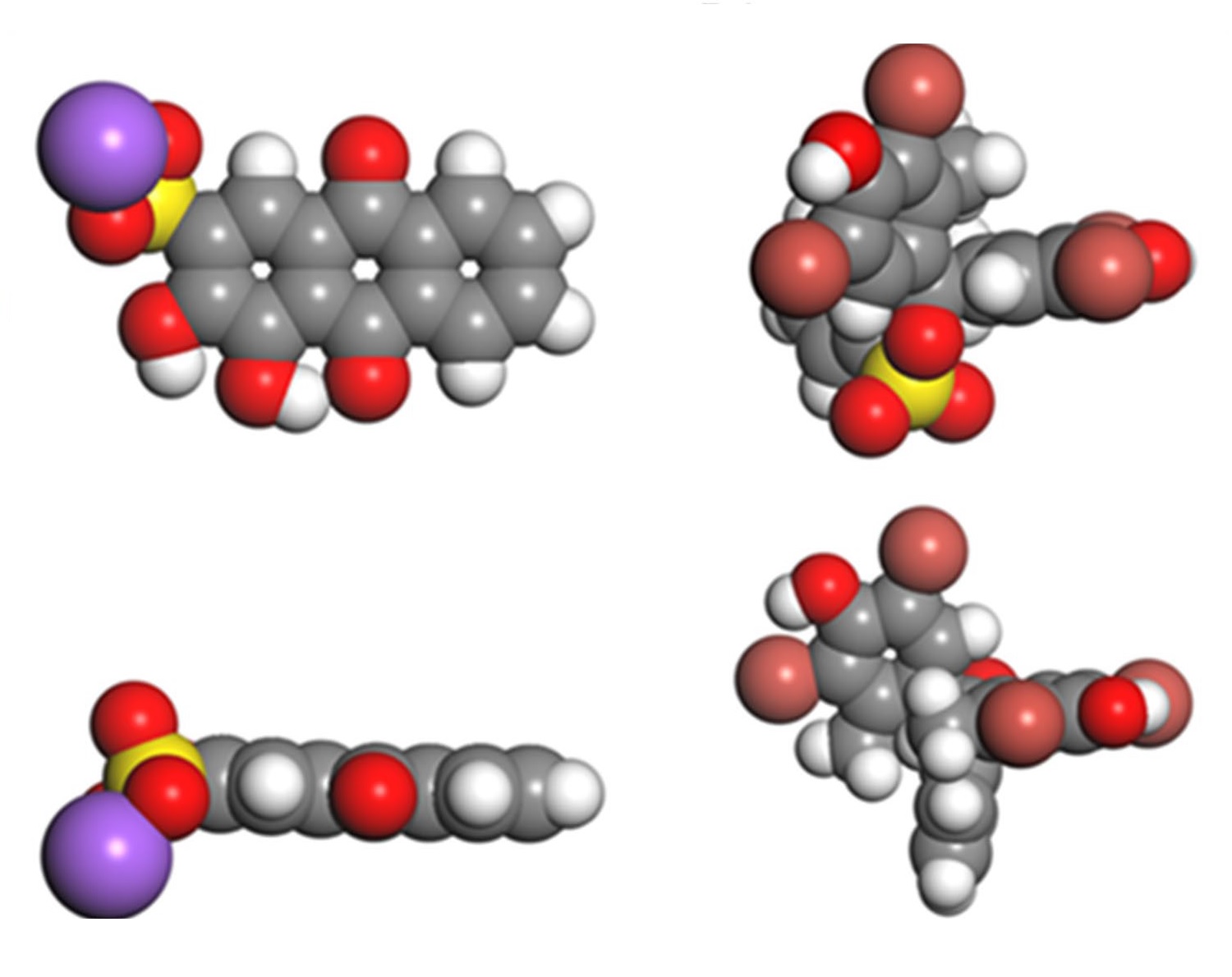
In this work, the effective adsorption and the subsequent photodegradation activity, of TiO2 brookite nanoparticles, for the removal of anionic dyes, namely, Alizarin Red S (ARS) and Bromocresol Green (BCG) were studied. Batch adsorption experiments were conducted to investigate the effect of both dyes’ concentration, contact time, and temperature. Photodegradation experiments for the adsorbed dyes were achieved using ultraviolet light illumination (6 W, λ = 365 nm). The single adsorption isotherms were fitted to the Sips model. The binary adsorption isotherms were fitted using the Extended-Sips model. The results of adsorption isotherms showed that the estimated maximum adsorption uptakes in the binary system were around 140 mg g−1 and 45.5 mg g−1 for ARS and BCG, respectively. In terms of adsorption kinetics, the uptake toward ARS was faster than BCG molecules in which the equilibrium was obtained in 7 min for ARS, while it took 180 min for BCG. Moreover, the thermodynamics results showed that the adsorption process was spontaneous for both anionic dyes. All these macroscopic competitive adsorption results indicate high selectivity toward ARS molecules in the presence of BCG molecules. Additionally, the TiO2 nanoparticles were successfully regenerated using UV irradiation. Moreover, molecular dynamics computational modeling was performed to understand the molecules’ optimum coordination, TiO2 geometry, adsorption selectivity, and binary solution adsorption energies. The simulation energies distribution exhibits lower adsorption energies for ARS in the range from − 628 to − 1046 kJmol−1kJmol−1 for both single and binary systems. In addition to that, the water adsorption energy was found to be between − 42 and − 209 kJmol−1kJmol−1.